Sage Therapeutics and Biogen Initiate Rolling Submission of New Drug Application (NDA) to U.S. Food and Drug Administration for Zuranolone for the Potential Treatment of Major Depressive Disorder (MDD) | Business Wire

Breakthrough Therapy, PRIME and Sakigake: A Comparison Between Neuroscience and Oncology in Obtaining Preferred Regulatory Status - Elena Tomaselli Muensterman, Yijia Luo, Jonathon M. Parker, 2019

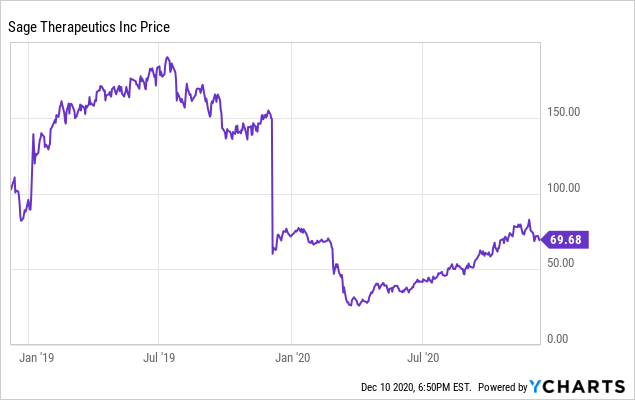
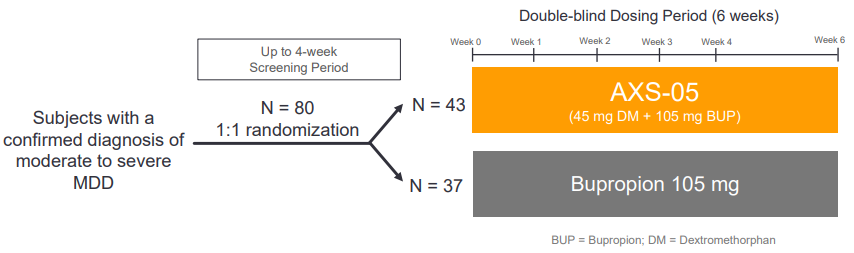
Sage Therapeutics Reports Topline Results from Pivotal Phase 3 MOUNTAIN Study of SAGE-217 in Major Depressive Disorder | Business Wire
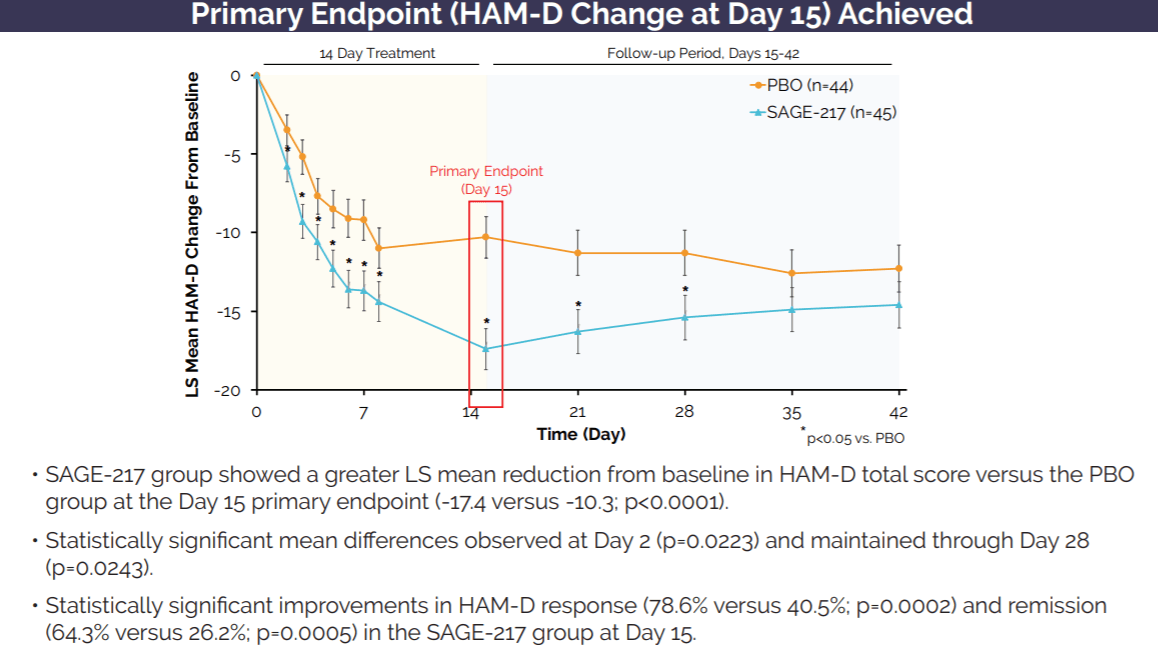
Sage Therapeutics: Zuranolone's Multi-Billion Dollar Market Opportunity More Than Justifies Company Valuation (NASDAQ:SAGE) | Seeking Alpha

Sage Therapeutics Reports Topline Results from Pivotal Phase 3 MOUNTAIN Study of SAGE-217 in Major Depressive Disorder
Sage Therapeutics Announces Second Quarter 2018 Financial Results and Highlights Pipeline and Business Progress

Sage Therapeutics Reports Results of SAGE-217 in P-III MOUNTAIN Study for Major Depressive Disorders
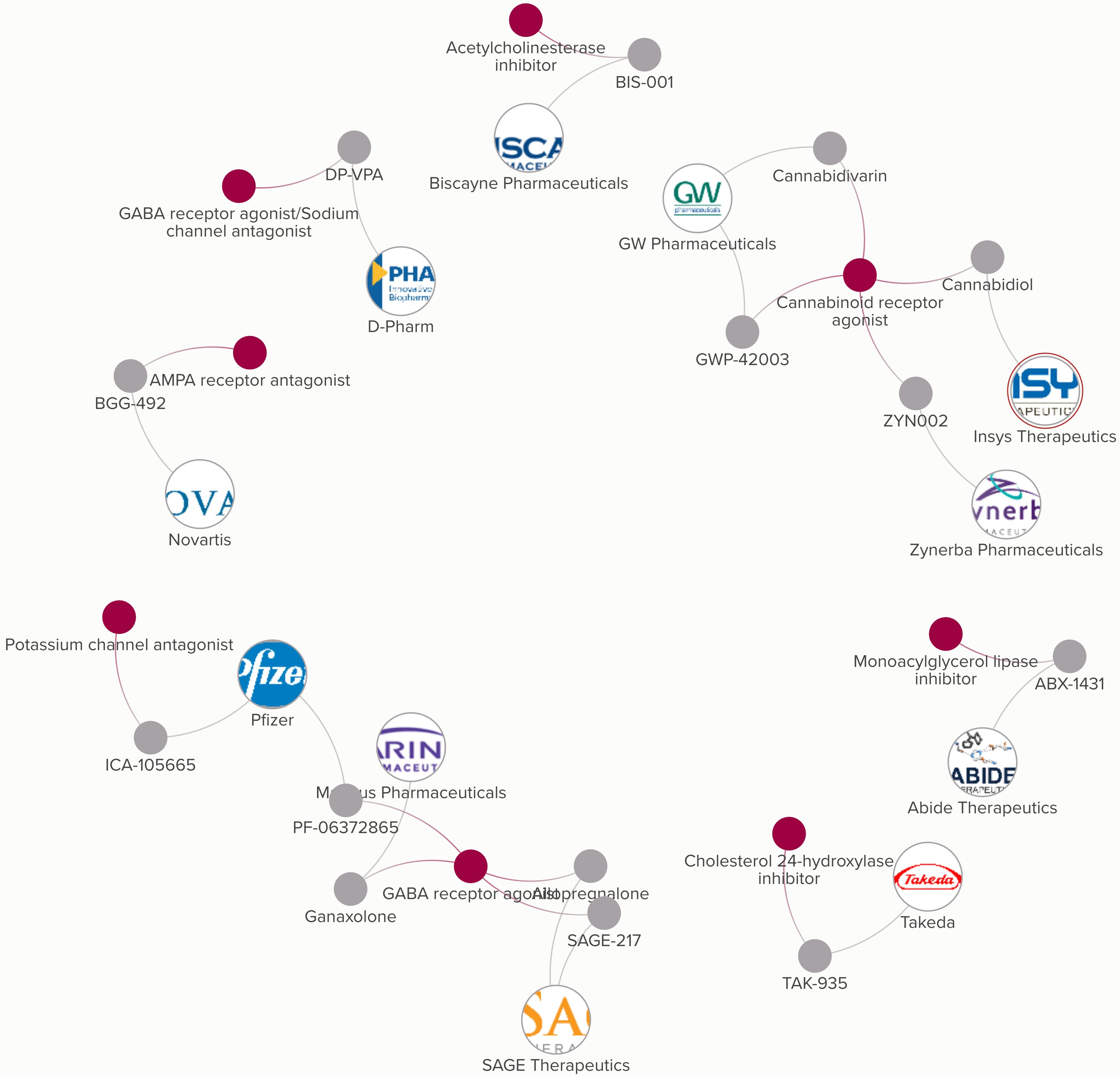
Defensive Strategies For Sage Therapeutics In The Aftermath Of Potential SAGE-547 Regulatory Action (NASDAQ:SAGE) | Seeking Alpha

Sage Therapeutics Announces Development Plan for Zuranolone (SAGE-217) Following Breakthrough Therapy Guidance Meeting with the U.S. Food & Drug Administration | Business Wire
Discovering, developing and delivering life-changing therapies to treat rare central nervous system disorders.