SOLVED: Question 42 Which amino acid is more conformationally restricted and why? proline because its side chain is bonded to both the nitrogen and the a-carbon atoms proline because its side chain

Which is the correct Lewis structure for carbon tetrabromide (CBr4), in which a central carbon atom - Brainly.com

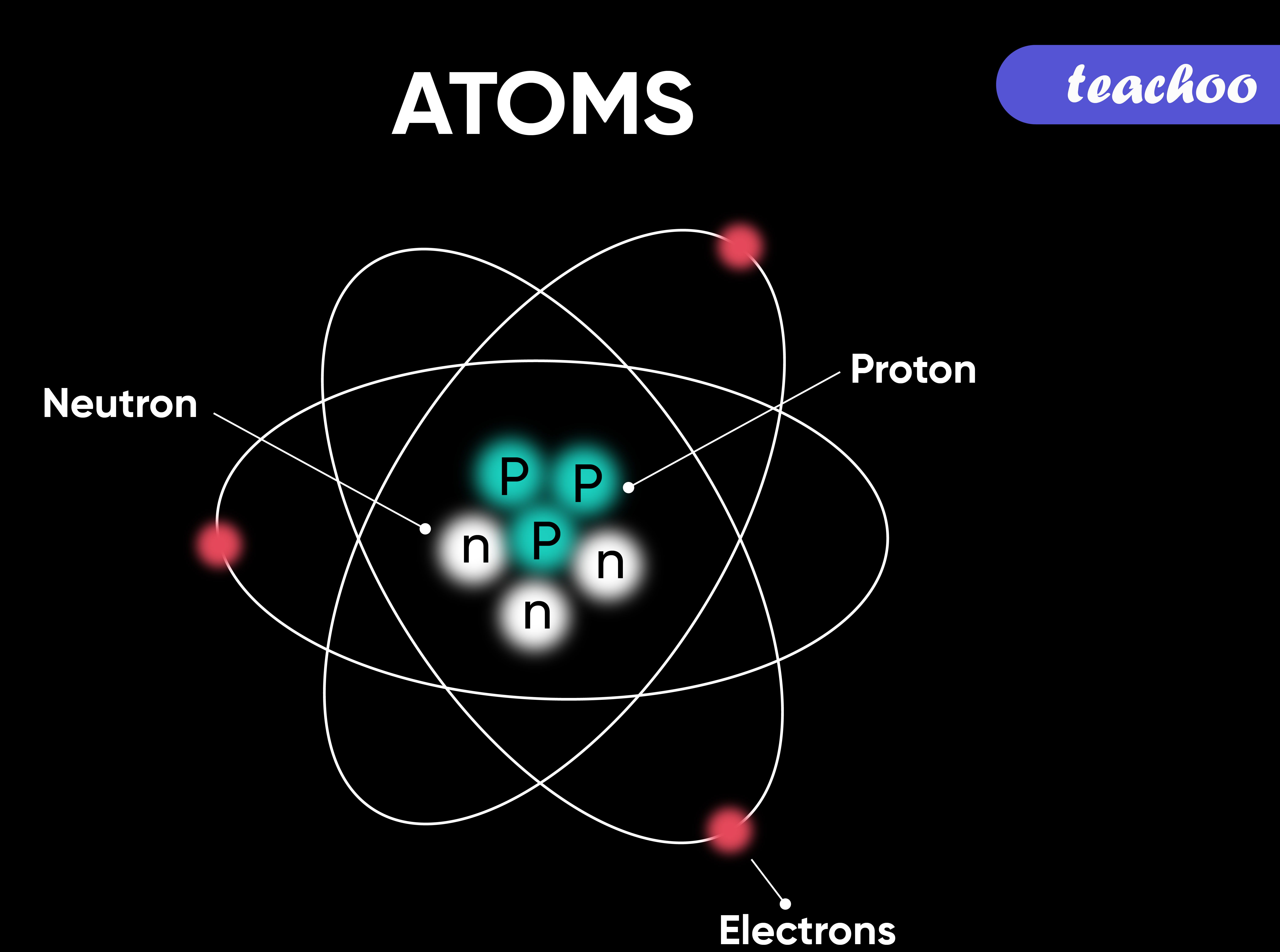
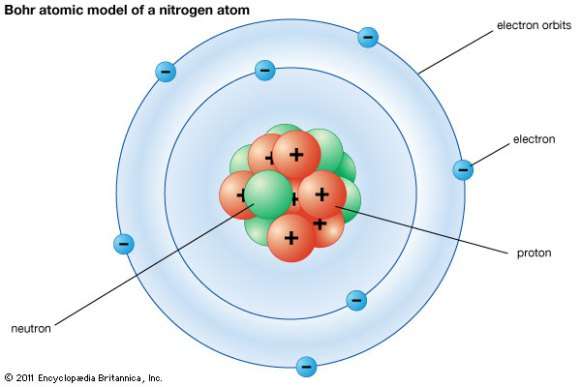
Q11 Draw the atomic diagrams of the following elements showing the distribution of protons neutrons ...

SOLVED: Name Use the Periodic Trends Worksheet periodic table and your knowledge of periodic trends t0 answer the following questions. Which atom in each 0) Li or K pair has the larger

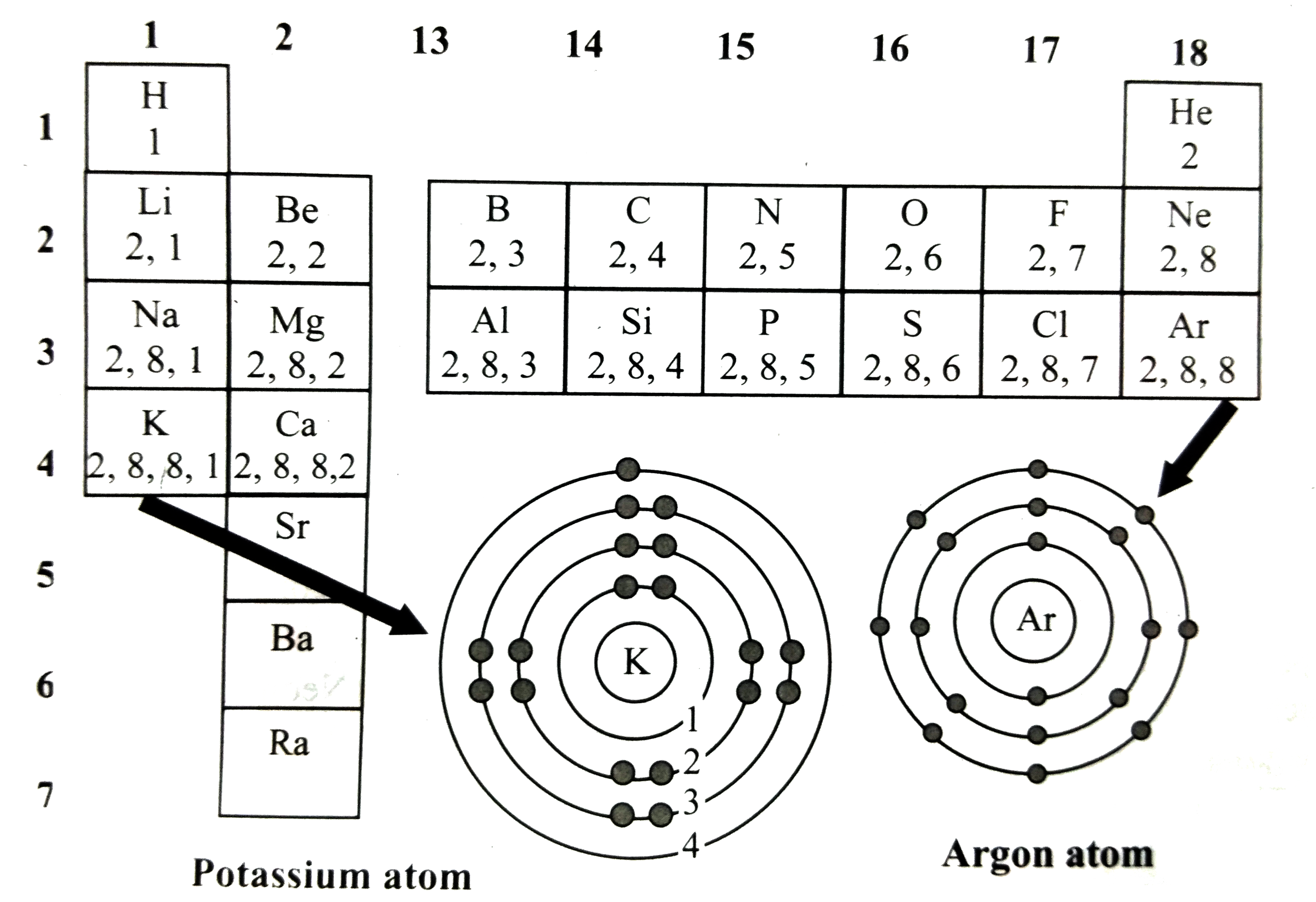
An atom has electronic configuration 2,8,7 .(a) What is the atomic number of this element?(b) To which of the following elements would it be chemically similar?(Atomic numbers are given in parentheses.)? N(7)

SOLVED: Nuclear Symbol Isotopic Mass 'Q7Ag 106.9 Percent Abundance 51.84% 109 47Ag 108.9 48.159/ (Show calculations here) Questions and Problems number of 80 and has 45 neutrons. Write its atomic svinbol: Q4

Gen Chem 1 Review Notes - Atomic Symbol onurged ion can gainlose electrons The Periodic Table · - Studocu